Lemon Cell Experiment
1. Aim
To construct a lemon cell and measure the EMF produced by different metal electrode pairs using one SEELab input channel.
2. Apparatus / Components Required
- SEELab3 or ExpEYES-17 unit
- Connecting wires / crocodile clips
- 1-2 lemons (or any acidic fruit)
- Common metal electrodes (for example: Zinc nail, Copper strip/coin, Iron nail, Aluminium strip)
- A PC, Laptop, or Android Phone with SEELab3 software
3. Theory & Principle
A lemon acts as an electrolyte. Two dissimilar metals inserted into the lemon form a galvanic cell.
The open-circuit voltage (EMF) depends on the electrode pair and electrolyte condition.
For this experiment, use one channel (A1 or A3) and measure with respect to GND.
Expected EMF trend follows metal reactivity difference:
- larger reactivity difference -> higher EMF
- copper is commonly used as the positive electrode in these pairs
Always measure voltage with respect to GND:
\(V_{\text{measured}} = V(\text{Input}) - V(\text{GND})\)
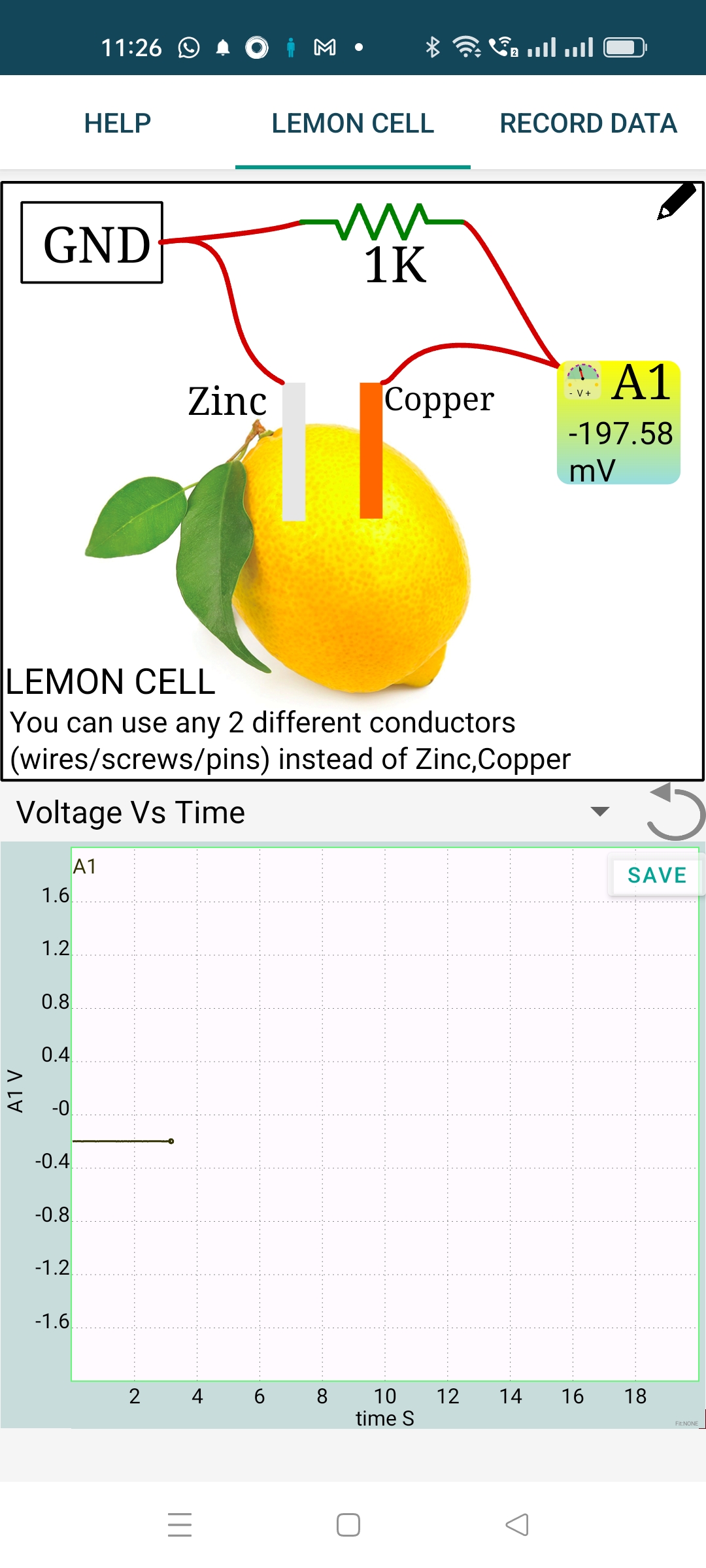
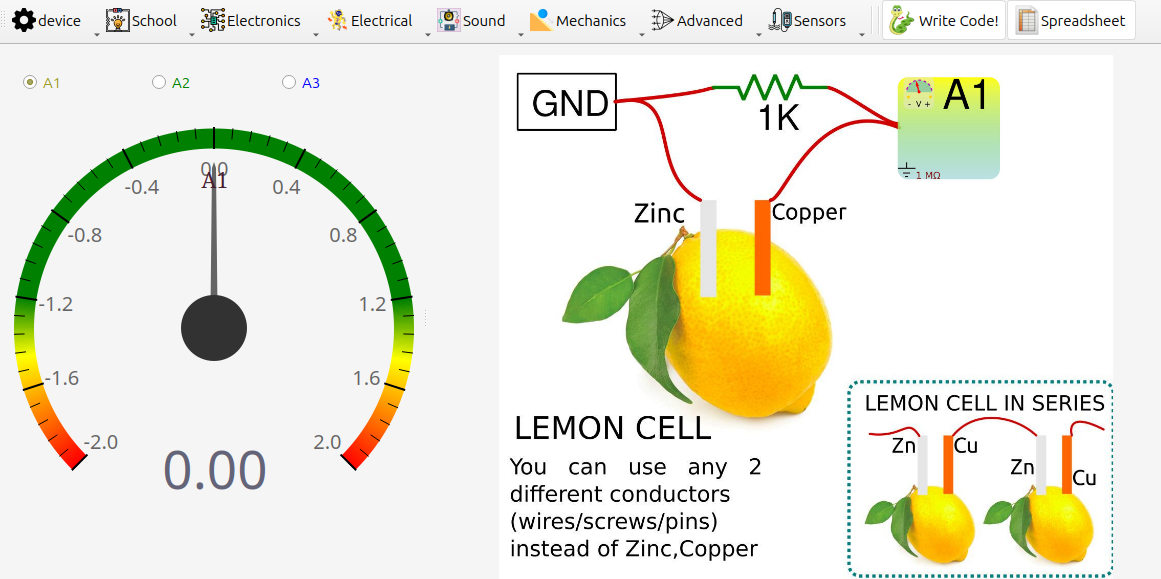
4. Circuit Diagram / Setup
- Select one input channel (
A1recommended;A3is also fine here). - Insert two different metal electrodes into the lemon, separated by ~2-3 cm.
- Connect the electrode expected to be negative (e.g., zinc) to
GND. - Connect the other electrode (e.g., copper) to the selected input.
- Open DC voltage measurement in the SEELab software/app.
5. Procedure
- Start with the Zn-Cu pair and note the voltage.
- Swap leads once and confirm sign reversal.
- Repeat with other pairs (Fe-Cu, Al-Cu, Zn-Fe, etc.).
- For each pair, wait 5-10 seconds for reading to stabilize and record value.
- Optional: connect two lemon cells in series and verify voltage addition.
Mobile App
Desktop App

6. Observation Table
| Metal Pair | Typical EMF Range in Lemon (V) | Measured Voltage (V) | Remarks |
|---|---|---|---|
| Zn-Cu | 0.8 to 1.1 | ||
| Al-Cu | 0.6 to 1.0 | ||
| Fe-Cu | 0.4 to 0.8 | ||
| Zn-Fe | 0.2 to 0.5 | ||
| Al-Fe | 0.1 to 0.4 |
7. Reference EMF Values (Common Metals)
Approximate standard electrode potentials (vs SHE, at 25 C):
| Metal / Electrode | $E^\circ$ (V) |
|---|---|
| Zn / Zn$^{2+}$ | -0.76 |
| Al / Al$^{3+}$ | -1.66 |
| Fe / Fe$^{2+}$ | -0.44 |
| Cu / Cu$^{2+}$ | +0.34 |
Approximate ideal EMF for common pairs (difference of $E^\circ$):
| Pair (Anode-Cathode) | Ideal $\Delta E^\circ$ (V) |
|---|---|
| Al-Cu | 2.00 |
| Zn-Cu | 1.10 |
| Fe-Cu | 0.78 |
| Al-Fe | 1.22 |
| Zn-Fe | 0.32 |
In real lemon cells, measured values are usually lower due to internal resistance, polarization, oxide layers, and non-standard ion concentrations.
8. Advanced: Estimating Internal Resistance of Lemon Cell
Model the lemon cell as an ideal source $E$ in series with internal resistance $r$.
When connected to a voltmeter of input resistance $R_{in}$, measured terminal voltage is: \(V = E\cdot\frac{R_{in}}{r+R_{in}}\)
For this experiment:
- A1 input resistance $R_{A1}\approx1M\Omega$
- A3 input resistance $R_{A3}\approx10M\Omega$
Steps
- Build one lemon cell (for example Zn-Cu).
- Measure open terminal voltage with A1: call it $V_{A1}$.
- Measure same cell with A3: call it $V_{A3}$.
- Use the formula below to estimate $r$.
From two readings: \(r=\frac{R_{A1}R_{A3}(V_{A3}-V_{A1})}{V_{A1}R_{A3}-V_{A3}R_{A1}}\)
Then estimate EMF: \(E=V_{A1}\left(1+\frac{r}{R_{A1}}\right)\)
Worked Example
Suppose measured values are:
- $V_{A1}=0.60V$
- $V_{A3}=0.90V$
Using $R_{A1}=1M\Omega$, $R_{A3}=10M\Omega$: \(r=\frac{(1)(10)(0.90-0.60)}{(0.60)(10)-(0.90)(1)}M\Omega =\frac{3}{5.1}M\Omega\approx0.588M\Omega\)
So internal resistance is about: \(r\approx5.9\times10^5\Omega\)
Now EMF: \(E\approx0.60\left(1+\frac{0.588}{1}\right)\approx0.95V\)
9. Error Analysis
Possible causes of small mismatch:
- Electrode surface condition: rust/oxide layer reduces effective EMF.
- Electrolyte variability: acidity differs from lemon to lemon.
- Internal resistance: lemon cell has high internal resistance, especially under load.
- Input loading: A3 (about $10M\Omega$) disturbs weak sources less than A1/A2 (about $1M\Omega$).
10. Results and Discussion
- Different metal pairs produced different EMF values.
- Pairs with larger reactivity difference generally gave higher voltage (e.g., Zn-Cu > Zn-Fe).
- For two lemon cells in series, total voltage approximately added: \(V_{\text{total}} \approx V_1 + V_2\)
11. Precautions
- Keep electrode tips clean for repeatable readings.
- Do not let electrodes touch each other inside the lemon.
- Use common
GNDreference and firm connections.
12. Troubleshooting
| Symptom | Possible Cause | Corrective Action |
|---|---|---|
| Reading near 0V | Same metal used / short / wrong wiring | Use dissimilar metals; recheck GND and input wiring |
| Unstable reading | Loose clips / dry lemon region | Reinsert electrodes, moisten area, tighten contacts |
| Too low voltage | Oxidized electrodes / weak acidity | Clean electrodes and try fresh lemon |
| Wrong polarity sign | Leads reversed | Swap electrode connections |
13. Viva-Voce Questions
Q1. Why does a lemon cell produce voltage?
Ans: Two dissimilar metals in an electrolyte form an electrochemical cell. Their different electrode potentials create an EMF.
Q2. Why is Zn-Cu usually higher than Zn-Fe in a lemon cell?
Ans: The electrode potential difference for Zn-Cu is larger than Zn-Fe, so EMF is generally higher.
Q3. Why are measured lemon-cell voltages lower than ideal EMF values?
Ans: Due to internal resistance, polarization, oxide layers, and non-standard chemical conditions.
Q4. If a 3V source is connected to A1 through a 1MΩ resistor, what is the displayed voltage (A1 input impedance = 1MΩ)?
Ans: Using voltage divider: $$ V_{A1}=3\times\frac{1}{1+1}=1.5V $$ Displayed value is approximately 1.5V.
Q5. For the same setup, what is displayed on A3 (input impedance = 10MΩ)?
Ans: $$ V_{A3}=3\times\frac{10}{1+10}=3\times\frac{10}{11}\approx2.73V $$ A3 shows approximately 2.73V.

